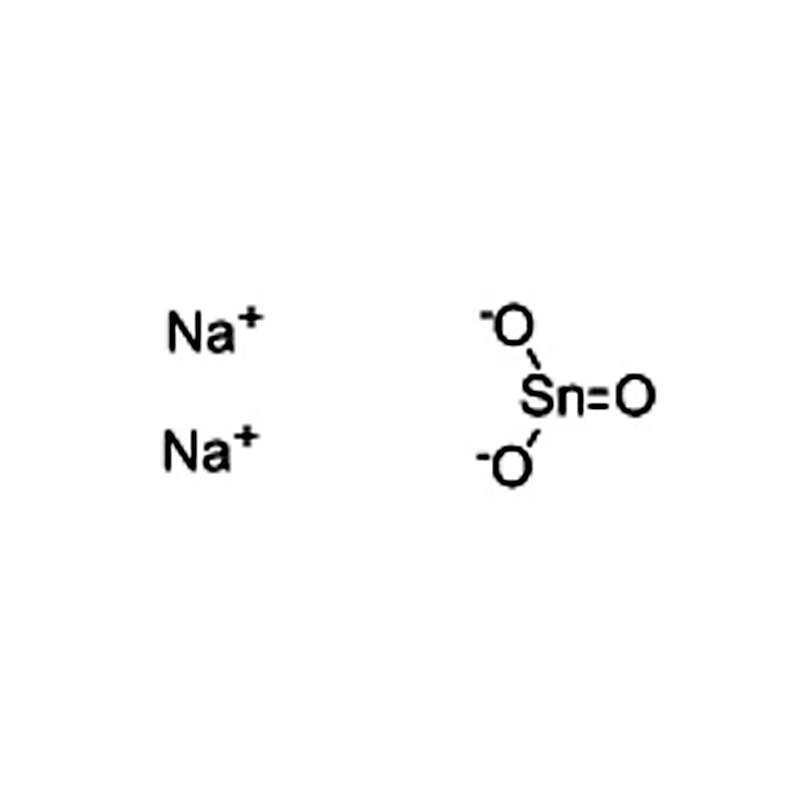
Sodium stannate CAS#12058-66-1
High Water Solubility: Easily dissolves in water, facilitating use in aqueous formulations and reactions.
Controlled Thermal Behavior: Loses crystallization water at 140 °C to form anhydrous sodium stannate, enabling predictable processing and handling.
Environmental Reactivity: Reacts with carbon dioxide from the air to form sodium carbonate and tin hydroxide, allowing versatile chemical applications.
Stable Physical Form: Available as colorless hexagonal plate-like crystals or white powder, providing consistent quality and ease of storage.
Sodium stannate is an inorganic compound, appearing as colorless hexagonal plate-like crystals or a white powder. It is soluble in water but insoluble in alcohol and acetone. Upon heating to 140°C, it loses its water of crystallization to form its anhydrous form. It absorbs carbon dioxide from the air to form sodium carbonate and tin hydroxide.
Sodium stannate Chemical Properties
| Melting point | 140°C |
| Density | 4.68 g/cm3(Temp: 25 °C) |
| Form | Crystalline |
| Odor | Odorless |
| Water Solubility | Slightly soluble in water. |
| Sensitive | Air Sensitive |
| Merck | 148677 |
| Exposure limits | ACGIH: TWA 2 mg/m3 |
| NIOSH: IDLH 100 mg/m3; TWA 2 mg/m3 | |
| Stability | Stable, but may be air sensitive. |
| Cosmetics Ingredients Functions | VISCOSITY CONTROLLING |
| LIGHT STABILIZER | |
| InChI | InChI=1S/2Na.3O.Sn/q2*+1;;2*-1; |
| InChIKey | TVQLLNFANZSCGY-UHFFFAOYSA-N |
| SMILES | [Sn]([O-])([O-])=O.[Na+].[Na+] |
| CAS DataBase Reference | 12058-66-1(CAS DataBase Reference) |
| EPA Substance Registry System | Stannate (SnO32-), disodium (12058-66-1) |
| Hazard Codes | Xi |
| Risk Statements | 36-36/37/38 |
| Safety Statements | 26 |
| WGK Germany | 3 |
| RTECS | JN6345000 |
| TSCA | TSCA listed |
Product Application of Sodium Stannate (CAS#12058-66-1)
Sodium stannate is used as a mordant in dyeing, as well as in ceramics and glass applications. It serves as a source of tin for electroplating and immersion plating, and is applied in textile fireproofing. Additionally, it functions as a stabilizer for hydrogen peroxide, is used in blueprint paper production, and serves as a laboratory reagent.